San Diego State University
Physics 197
tonydude@yahoo.com
Help Page Syllabus
Test Questions 3,4, Final
Chapters. . (16, 17, 18, 34), (35, 36, 37, 38, 39), (40, 42, 43, 44, 45, 46)
Chapter 37-Interference
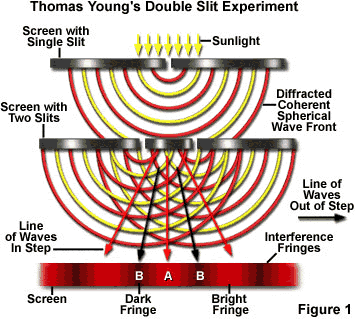
Young's Double-Slit Experiment (Wave Nature of Light)
One of the most important experiments of wave theory is that of Thomas Young's double slits. It is a clear example of the diffraction of light conducted with essentially basic scientific equipment. Thomas Young was a not only a physicist but also a physician and and Egyptologist, who was responsible for deciphering the Rosetta stone. He devised an experiment in the early 1800's that proved that light is a wave. The experiment has been used subsequently to show that wave behavior exists in many other areas of nature and therefore it is worth spending a little time going into the experiment in detail.
courtesy of: Molecular Expression
Double-Slit Theory

HyperPhysics

Tony's Double-Slit Notes

First Run
Without Diffraction
With Diffraction
N-slits
Intensity

Tony's Intensity Notes

Waves or Particles?
Young or Newton?
"It was inconceivable that particles of light coming through the slits
could cancel each other in a way that would explain the dark fringes."
Light is most certainly a wave phenomena according to this experiment!
Phasors
Thin Film Interference


Thin Film Calculation-HyperPhysics
Tony's Notes

Diffraction Pattern around a Penny!
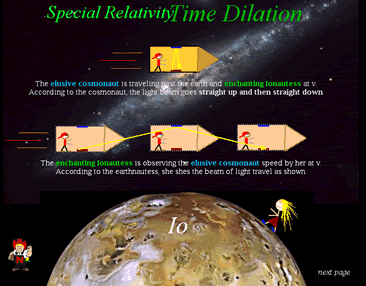
Chapter 39-Special Relativity(<---don't click there)
You are traveling at 50 mph in a pick up truck.
You throw a ball at 50 mph to someone on the side of the road.
What speed is the ball traveling when the person on the road catches it?
We all know that one would simply add (or, subtract) the two speeds. . .right?

This is what Galileo presumed . . .

The Laws of Mechanics are the same in all inertial reference frames.
Looking for the elusive Aether.
How does light travel through ‘empty’ space?
Is space empty?
What does a vacuum really mean?
No, it does not mean you have to clean the house!
Step up Michelson and Morely. . .or here


You want to derive the Equations of Relativity?
Can anyone prove this nonsense?
Muons?


Chapter 41-Quantum Mechanics
Quantum Mechanics is the study of matter and radiation at an atomic level.
Intro to Quantum Mechanics
Electron (Matter) Waves!

Electrons demonstrate Interference. Electrons are Waves?
Actually, light and matter exist as particles; what behaves like a wave is the probability of where that particle will be. The reason light sometimes appears to act as a wave is because we are noticing the accumulation of many of the light particles distributed over the probabilities of where each particle could be.
The WaveFunction?
David Griffith's Interpretation

Electrons act like particles when we are looking. . .and
like waves when we are not looking!
It is Physically impossible to measure simultaneously the
exact position and exact linear momentum of a particle
At the atomic scale of quantum mechanics, however, measurement becomes a very delicate process.
Let's say you want to find out where an electron is and where it is going. How would you do it?
Get a super high powered magnifier and look for it?
The very act of looking depends upon light, which is made of photons, and these photons could have enough momentum that once they hit the electron they would change its course!
It's like rolling the cue ball across a billiard table and trying to discover where it is going by bouncing the 8-ball off of it; by making the measurement with the 8-ball you have certainly altered the course of the cue ball. You may have discovered where the cue ball was, but now have no idea of where it is going (because you were measuring with the 8-ball instead of actually looking at the table).

Particle in a Finite Box
Games physicists play. . .
the torture box. . .don't ask

Could this be an electron in an atom?
Classically the particle (electron) is permanently bound.

However, Quantum Mechanics insists that there is a probability
that the particle (electron) can be found outside the well!
Whatever. . .

Barrier Penetration---Tunneling
Alpha and Beta Radiation
Popular Mechanics articl


Even Schrödinger was unhappy with the implication of his findings.
"I'm sorry that I ever had anything to do with quantum theory,"
he is said to have lamented to a colleague.
One of the principal consequences of uncertainty is that you cannot specify the exact state of a particle without somehow interacting with it.
Chapter 43-Solid State Physics
Chapter 44-Nuclear Structure



A Simple Calculation



![]()
The History of Radioactivty


More info



Tunnel Theory of Alpha Decay



Disintegration Energy

Chapter 45-Nuclear Fission and Fusion
Chapter 46-Particle Physics and Cosmology
Test #3
INTERFERENCE
Test Question #1
Using a diagram, describe or explain how Young utilized the Double-Slit to discover that light travels in waves.
Please be detailed in your explanation and your diagram.
How was it possible that Young was able to measure the very small wavelengths of light?
Grading: Diagram, Equations, Explanation for wave theory and how to find wavelength.
or
Using a diagram, describe or explain how Michelson used his Interferometer to measure the
index of refraction of a gas in a cylinder. Please be detailed in your diagram and explanation.
Grading: Diagram, Equations, Explanation for wave theory and how to find index of refraction.
(20 points)
You will choose one of the above two for the test.
Solution
INTERFERENCE and DIFFRACTION
Test Question #2
Chapter 37---#34,39,59, or 60 (I will change numbers but, not the problem)
Chapter 38---#15,28, or 33 (I will change numbers but, not the problem)
(20 points)
I will choose one of the above for the test.
Solution
RELATIVITY
Test Question #3
Chapter 39---#8,9,15,21,23,25,38,48,57, or 73 (I will change numbers but, not the problem)
(15 points each)
I will choose two of the above for the test.
Solution
Solution
QUANTUM PHYSICS
Test Question #4 and #5
Chapter 40---Explain using Diagrams, Equations, Words and Logic. . No derivations necessary!
1) How did the idea of Blackbody Radiation lead to the Ultraviolet catastrophe and Planck’s Hypothesis?
2) How did the Photoelectric Effect demonstrate that the classical wave theory of light is wrong and that the
quantum nature of light was correct? Demonstrate by using #65.
3) How did the Compton Effect demonstrate that the classical wave theory of light is wrong and that the
quantum nature of light was correct?
4) How does de Broglie's Theory and the Davisson-Germer experiments resolve the conflict between
the particle theory and the wave theory? Demonstrate by using #57
(15 points each)
You will choose (1 and 3) or (2 and 4) of the above for the test.
Solution
Solution
Good Luck, everyone.
Test #4
Chapter 41 - 12,38,53
Chapter 44 - 29,34,38,62
Chapter 45 - 9,16,43,56
Chapter 46 - 31,37,47
DiMauro's Final
Due Monday, July 7, 2003
1) Derive the Lorentz Transformation Equations (39.11) (Hints: 1,2)
or, Alternatively. . .Chapter 39 #65
Please be detailed as needed and explain each step in words.
(25 points)
2) Create Figure 44.8, using the semiempirical mass formula. (Hints:1,2,3)
I want you to re-create this graph, using excel. You will turn in a graph as well as a chart.
Please do NOT copy your friends graph or chart. I would like to see a table. Please provide a short explanation.
Use the Hints to guide you.
First, I plotted the Volume term.
Second, I plotted the Volume term - Surface term.
Third, I plotted the Volume term - Surface term - the Coulomb repulsion.
.
.
.
I want you to play with the graph. Demonstrate that you know
what is going on.
It does NOT have to be exactly like mine. Use your imagination.
I will grade the graph and data based upon: Completeness, Clarity and
Composition
(25 points)